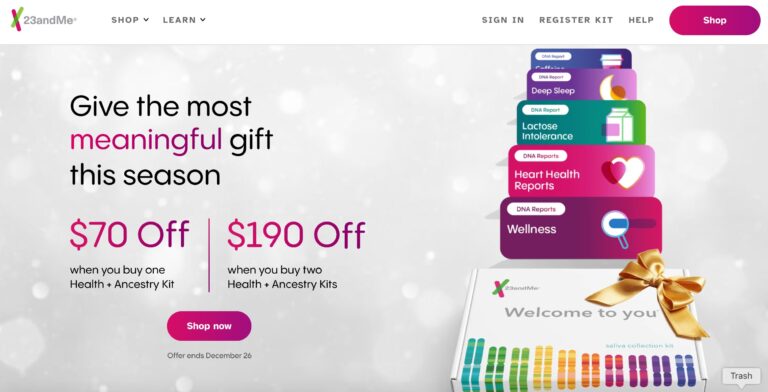
When it comes to consumer genetics testing, 23andMe ranks at the top. Here, we give...
Read MoreWelcome To USA Rx
Trusted Medication Resources in the U.S.
Trusted Resources by Pharmacists in the U.S.
Save Up to 70% Today on Your Prescription Medications.
Home Health Reviews for 2024
The Best Online Doctor and Medical Services in 2024
The best online doctor and medical services in 2023; include industry staples like Teladoc and...
Read MoreHairClub Reviews: Are These Hair Loss Experts Legit?
For years, HairClub's been offering lifelines for those with hair loss, but are their hair...
Read MoreWalgreens App Review: Deliveries, Medicine Refills, & More
Walgreens is one of America's leading pharmacy chains, but does their app meet the expectations...
Read MoreK Health Review: Is This AI-Based Medical Care Legit?
K Health is shaping the future of healthcare with its affordable pricing and innovative approach;...
Read MoreEverlyWell Review: What You Need To Know Before You Order a Test
Everlywell is a telehealth company that offers affordable at-home testing kits for tons of health...
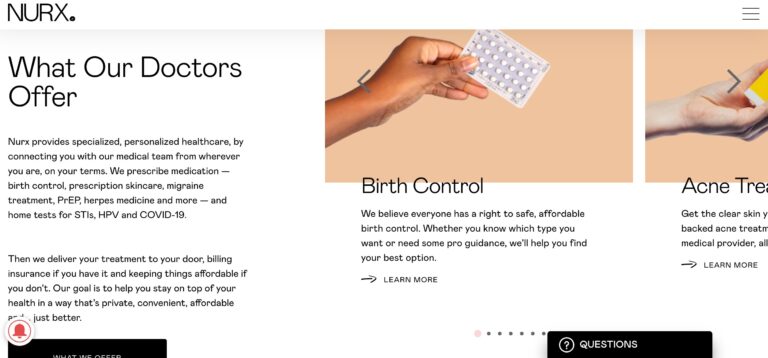
Read MoreNurx Review: Why Their Personalized Healthcare Is Unique
Nurx is a telemedicine company that could help you get medications easier than visiting your...
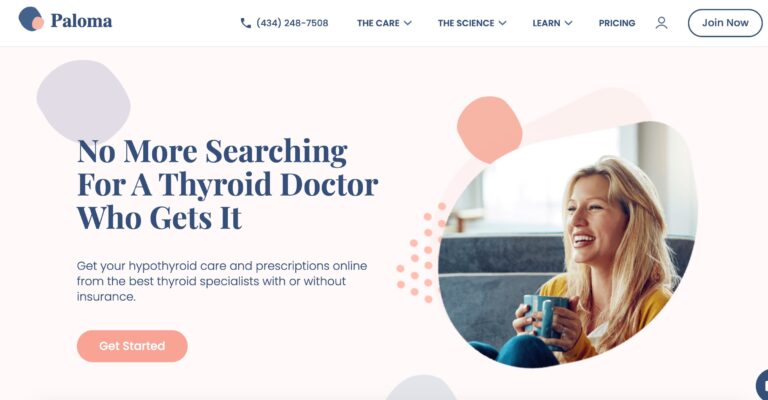
Read MorePaloma Health Review: Is This At-Home Thyroid Test Effective?
Paloma Health's at-home thyroid test offers an easy way to diagnose potential thyroid conditions; but...
Read MoreMyLabBox Review: Are These At-Home Tests Legit?
At-home testing for STDs and other health concerns is growing increasingly popular, but myLAB Box...
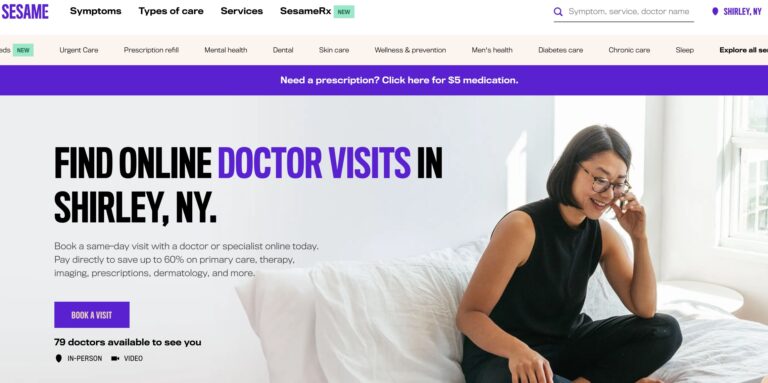
Read MoreSesame Telemedicine Review: Are These Online Doctor Visits Legit?
Sesame is a direct-to-consumer telemedicine company that offers a variety of services for a single,...
Read MoreThe Best Hair Loss Treatment for Men: Comparison Guide
If you’re looking for the best hair loss treatment for men, look no further than...
Read MorePicnic Allergy Review: Does This Personalized Treatment Work?
Picnic is a telehealth company that provides personalized treatment for allergy relief, but do they...
Read MoreNutrafol Review: Are These Hair Supplements Effective?
Nutrafol is a telehealth company that offers a range of hair supplements for both men...
Read MoreTeladoc Reviews: Pros & Cons of Virtual Health Counseling
Teladoc is a direct to consumer healthcare company that offers medical care online or via...
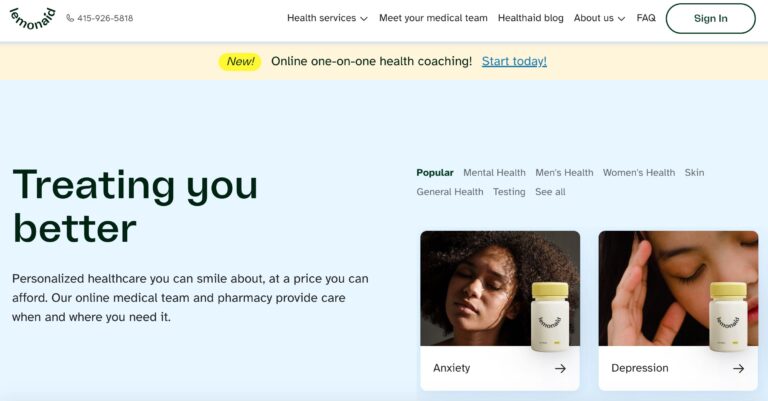
Read MoreLemonaid Health Reviews: What To Know Before You Buy
Lemonaid Health is one company looking to bring affordable telemedicine to all. Here are a...
Read MoreAt-Home STD Test Reviews: Complete Comparison Guide
At-home STD test options range from fully online options to tests that require a visit...
Read MoreShapiro MD Review: Does Their Hair Loss Treatment Work?
Shapiro MD uses natural DHT blockers in its shampoos, conditioners, and topical treatments, which may...
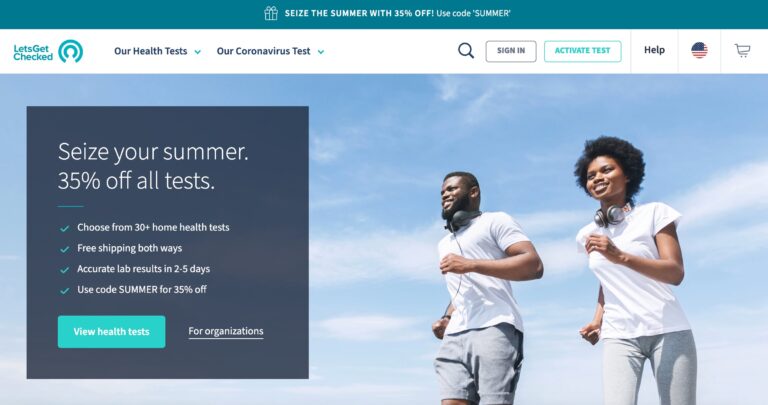
Read MoreLetsGetChecked Review: Are Their Home Health Tests Legit?
Considering at-home testing? LetsGetChecked is a great resource. Learn how this company works and what...
Read MoreMeet Our Medical Team
USARx.com is a trusted resource for medications and medical writing.
Our team of pharmacists and medical writers are dedicated to providing accurate, up-to-date information on medications and their side effects.
We understand that deciding which medication to take can be a difficult and confusing process, so we aim to make it as easy as possible for you to find the information you need.
Our website provides detailed descriptions of the most commonly prescribed medications, as well as articles on a wide range of topics related to medication use and safety.
In addition, our team is available to answer your questions via email or live chat. Whether you’re looking for information on a specific medication or just want to learn more about how medications work, we’re here to help. Thanks for choosing us!
Medication, Pharmacy, and Health Research and Statistics
Pharmacy Deserts
Exploring Pharmacy Density Nationwide
Read MoreVaccine Passports
We’ve surveyed over 1,000 people to get their takes on this latest political controversy surrounding...
Read MorePerceptions of Marijuana and Psychedelics
Let’s take a closer look at how many Americans report using different drugs (including marijuana,...
Read MoreThe States That Lie to Their Doctor the Most
Have you ever lied to your doctor? If your answer is yes, don’t worry, you’re...
Read MoreSurveying Americans: Items You Have to Pay for but Should Be Free
Shouldn’t vaccinations, ambulance rides, and diapers be free? We surveyed 1,000+ Americans to uncover which...
Read MoreAI Gives Medical Advice — Survey Says..
To determine if people trust AI for medical and health advice, we surveyed 1,015 participants...
Read More